
HSPs form oligomeric complexes constitutively expressed under normal conditions (ranging up 5-10% of the total protein). Then, it was noted that HSPs are expressed in a highly conserved manner in all organisms from bacteria to humans, to the point that many times they may be efficiently interchanged. The biological consequence was an exceptional increase of transcriptional activity for a family of proteins that, for this particular reason, were generically named heat-shock proteins (HSPs). One of his colleagues accidentally switched the temperature of his incubator where Drosophila cells were grown, and a quite different puffing pattern was acquired by those cells. The stress response to heat-shock was originally described in the early ‘60s by the Italian researcher Ferruccio Ritossa. The generation of a new proteostasis network implies an immediate role in protein synthesis, folding, disaggregation, or degradation, processes that encompass the translational machinery, molecular chaperones and their associated cochaperones, the ubiquitin-proteasome machinery, and the autophagy system. Importantly, such response builds a physiological network that shields cells and tissues from the risky challenges they may encounter, such as heat, cold, toxics, chemicals, UV light, radiation, drugs, infections, infestations, inflammation, pH variations, osmotic changes, nutrient deprivation, oxidative stress, hypoxia-ischemia, apoptotic stimuli, and stressful conditions for individuals such as psychiatric disorders and socially traumatic experiences. With the purpose to protect proteins from misfolding, denaturation and/or aggregation, cells trigger a fast response characterized by a number of events able to protect them from the hostile environment, and restore a balanced and safe new steady-state of protein homeostasis commonly referred to as ‘proteostasis’. Introduction to the stress responseĬells are always exposed to a number of sudden and potentially harmful variations of their biological milieu. In this article we review the basics of the stress response, summarize current controversies over the role of extracellular chaperones in inflammatory reactions and autoimmunity, and discuss the cytoprotective and immunoregulatory roles of heat-shock proteins, a challenging subject that may open a new avenue for the drug discovery and treatment of diseases related to autoimmune disturbs. Therefore, it has been proposed that immunization with heat-shock protein peptides could prevent the development of certain diseases. These extracellular chaperones are capable to stimulate anti-inflammatory regulatory T cell responses, thereby inducing the negative feedback control of inflammation. In several inflammatory pathologies, chaperones are greatly induced as a direct consequence of the inflammatory stress and are released from the cell thanks to a poorly understood mechanism. 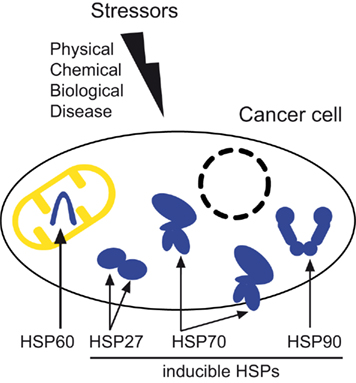
In addition to numerous components in immune responses, chaperone proteins are also detected in the extracellular fluids and have been implicated in autoimmune and inflammatory diseases acting as pro- and anti-inflammatory factors. It is currently accepted that misdirected immune responses may target self-antigens and generate severe inflammatory responses, a typical signature of autoimmune diseases. They are also active players in immunologically relevant scenarios such as the activation of innate immunity, antitumour immunity, and autoimmune diseases. Classically, molecular chaperones play a pivotal role in the maintenance of cellular proteostasis and thus, in the safeguarding of the cell homeostasis while reducing the deleterious effects of extracellular and intracellular stresses.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
